FDA Changes Label Requirements
Why did the FDA make changes to label requirements?
The U.S. Food and Drug Administration wants to make sure consumers have updated nutritional information for packaged foods sold in the United States. This will help people make informed decisions about the foods they eat and feed their families. The new label is a vast improvement and helps consumers can make more informed food choices to better their health.
What you need to know about the FDA’s new Nutrition Facts panel.
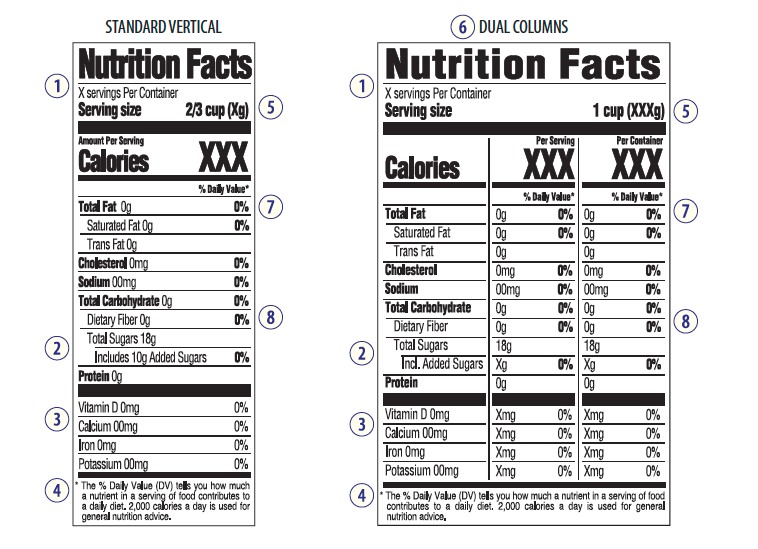
The calorie count is a bigger font and easier to find, serving sizes are more “realistic” in relation to the actual services per container, and the total sugars will include any added sugars in processing on a separate line (using the word “included” to show the second line is still a part of the total sugars already called out). There will also be a percent daily value (%DV) for added sugars.
Most manufacturers will be required to make changes to the nutrition facts panel by July 26, 2018. Manufacturers with less than $10 million in annual sales have one additional year (July 2019).
So what are some of the changes you should be aware of?
1. Serving Size: Examples: a 20oz bottle of soda may have to calculate a service as “per bottle” and not just as an 8oz service. Or, a bag of chips will have to show a more reasonable service than “8 chips”, when most people consume more than just 8 chips. These changes are to better show the service size and reference amounts customarily consumed [RACCs].
2. Added sugars – with % DV.
3. New requirements for nutrients. (Vitamin D and potassium values will replace vitamin A and vitamin C.)
4. Updated footnote (simplified and should better explain % of Daily Value in a daily diet.)
5. Graphic updates that highlight Calories and Service Size information. (“Calories from Fat” are removed.)
6. Duel-column Nutrition Facts panel on food listing “per serving” and “per package” (foods that could be consumed in one or multiple sittings).
7. Updated calculations for Daily Values and reference amounts (including sodium, dietary fiber, cholesterol and carbohydrates).
8. Dietary fiber (Definition is being updated by the FDA and a separate notice will be published.)
We can help!
Immotion Studios can help you with food labeling and regulatory review. Email lhurr@immotionstudios for more information.
For a full copy of all regulations, refer to the FDA’s federal regulations.
Written by Patti Bandy, Art Director